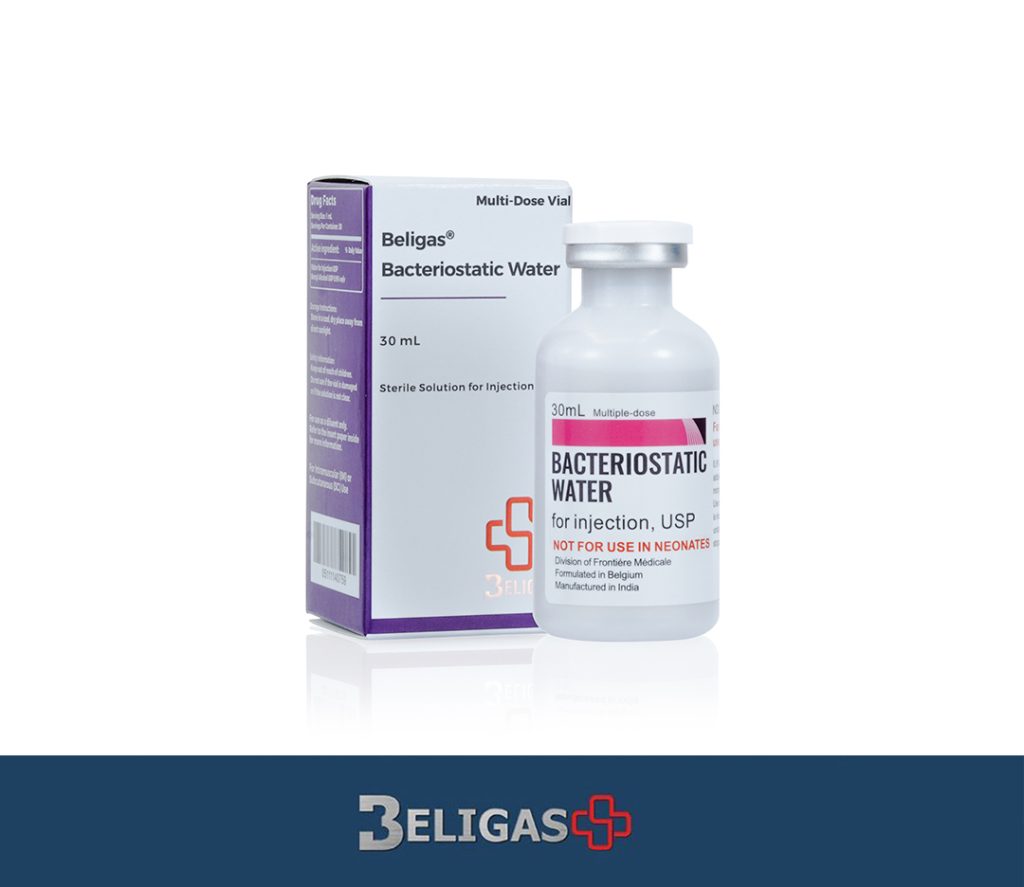
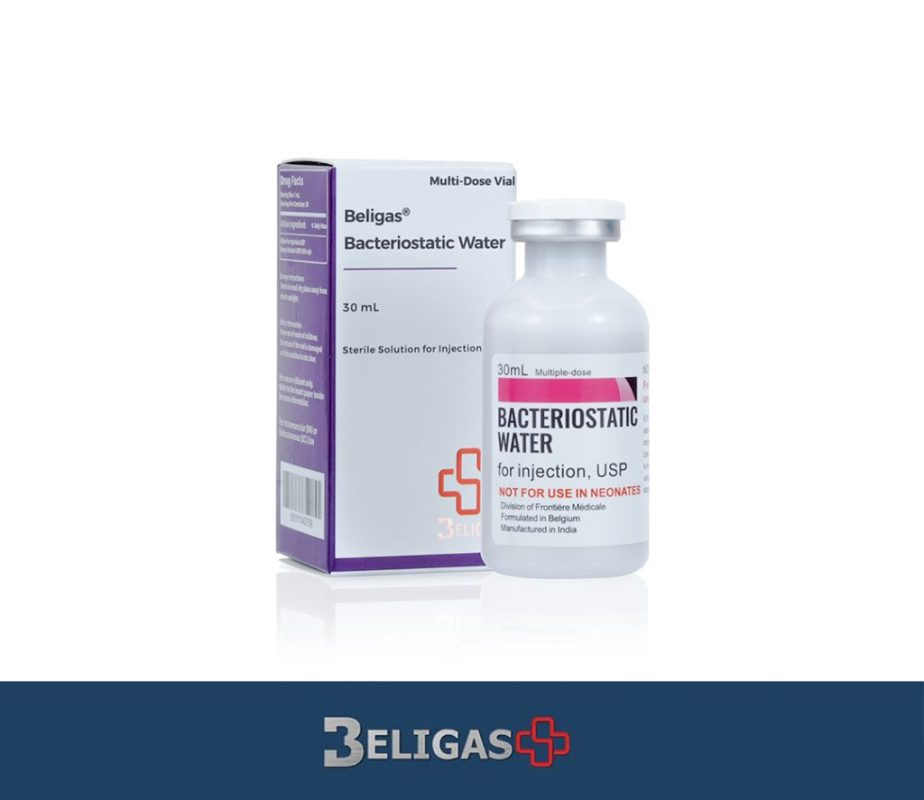
Bacteriostatic Water, 30ml (1 vial)
Brand Name: Beligas
Ingredient: Bacteriostatic Water
Dosage: 30Ml
Description:
Bacteriostatic Water is a sterile, non-pyrogenic aqueous solution used as a diluent or solvent for the preparation of injectable medications. is specially designed to inhibit the growth of bacteria and is commonly used in healthcare settings for reconstituting powdered medications, including vaccines and various injectable drugs. Bacteriostatic Water is typically available in 30mL vials, providing a sufficient volume for multiple uses while maintaining sterility.
Indications:
The primary indication for Bacteriostatic Water is as a diluent for reconstituting medications that will be administered via injection. It is utilized for preparing injectable solutions from powdered forms of drugs, allowing for accurate dosing and administration. Additionally, Bacteriostatic Water may be used in various clinical scenarios requiring a sterile vehicle for injection.
Uses:
- For Reconstitution of Medications: Bacteriostatic water is often used to dilute or dissolve medications for injection. The dosage of the medication will vary depending on the specific medication being reconstituted. Always follow the manufacturer’s instructions or a healthcare professional’s guidance for the appropriate volume of bacteriostatic water to mix with the drug.
- General Volume: For preparing injectable solutions, a common practice is to use between 1 mL to 5 mL of bacteriostatic water for reconstituting powder medications. Again, consult the specific medication’s guidelines.
Precautions:
While Bacteriostatic Water is generally considered safe for use, certain precautions should be observed. It is essential to check for any particulate matter or discoloration before use. Moreover, Bacteriostatic Water should not be used in neonates, as benzyl alcohol can be associated with a toxic syndrome known as “gasping syndrome” in this population.
Storage:
After opening the vial of bacteriostatic water, it can typically be stored at room temperature for up to 28 days. However, proper storage conditions must always be followed to maintain sterility and efficacy.
Regulatory Compliance Statements:
- This product complies with relevant regulatory guidelines.
- Certified under Good Manufacturing Practices (GMP).
Safety Information:
- Overdose Instructions: In case of overdose, seek immediate medical attention.
- Adverse Reactions: Report any adverse reactions to your healthcare provider.


Reviews
There are no reviews yet.